module 16
Herbicide resistance in weeds
Objectives
This Module is designed to introduce the concept of herbicide resistance, in all its forms, with an emphasis on the problems it causes in Australian agriculture. At the end of the topic you will:- understand the concept of herbicide resistance;
- be familiar with how weeds become resistant to herbicides;
- appreciate how resistance can be spread;
- be able to explain the situation within Australia and Queensland with respect to herbicide resistance; and
- describe some of the steps that can be taken to help prevent the future evolution of herbicide resistance in other populations of weeds.
Module outline
- What is herbicide resistance?
- How does a weed become resistant to a herbicide?
- Factors influencing the development of resistance
- Herbicide resistance in Australia
- Weed species at risk
- Current status of herbicide resistance in the Northern Grain Region
- The future
- References
Herbicide resistance
(Modified from the CRC for Australian Weed Management’s ‘Integrated Weed Management Manual’)
Herbicide resistance is defined as:
the genetically inherited ability of a plant to survive application of a herbicide that is normally lethal to the vast majority of that species.
Herbicide resistant weed populations are now found throughout all cropping areas of Australia from Western Australia to central Queensland. Currently, 25 weed species in Australia have been identified as being resistant to one or more herbicide mode-of-action (MOA) groups. The number of resistant species and areas affected by resistance will continue to increase until integrated weed management practices are widely adopted in Australian cropping systems.
Herbicide resistance fact box (an update from earlier courses):
- resistance is the inherited ability of an individual plant to survive a herbicide application that would kill a normal population of the same species;
- seven distinct MOA groups are no longer effective against one or more resistant weed populations;
- herbicide resistance is normally present in very low frequencies in weed populations before the herbicide is first applied. Variation exists within every population, with some individuals having the ability to survive the herbicide application;
- the frequency of resistant individuals within a population will vary greatly within and between species and MOA groups;
- a weed population is defined as resistant when a herbicide that once controlled the population is no longer effective (sometimes an arbitrary figure of 20% survival is used);
- the proportion of herbicide resistant individuals will rise (due to selection pressure) in situations where one herbicide MOA group is applied repeatedly;
- herbicide resistance is permanent in weeds and their progeny with dominant target-site resistance. With cessation of the use of that herbicide MOA group, the ratio of dominant target-site resistant to susceptible individuals will remain the same – only the total number of weeds present can be reduced. Weeds with this type of resistance do not exhibit a fitness penalty.
What is herbicide resistance?
It is important to differentiate between herbicide resistance and herbicide tolerance.
Herbicide resistance is the inherited ability of a plant to survive and reproduce following exposure to a dose of a herbicide normally lethal to the wild type (Heap 2006).
Herbicide resistance does not equate to poor performance of a herbicide. Resistant weeds can often survive application of herbicide at rates that are much greater than the recommended rate.
Herbicide tolerance is the inherent ability of a species to survive and reproduce after herbicide treatment at a normal use rate (Heap 2006).
There is no selection involved (through herbicide application) because the species is naturally tolerant. Examples of herbicide tolerance include wheat or Vulpia spp. to post-emergent applications of diclopfop (Group A).
Herbicide tolerance is the underlying feature of herbicide selectivity. It enables crops to survive applications of selective herbicides and explains why selective herbicides do not kill all weeds.
How does a weed become resistant to a herbicide?
There are three major ways in which resistance may arise within a weed population:
Pre-existing resistance
Within any weed population there may be some plants that already contain a rare change in a gene (or genes) that enables them to survive the application of a particular herbicide that would normally kill this species.
Genetic variation may alter physiological traits that enable herbicide uptake, translocation and activation at the site of action. Alternatively, changes may influence the plant’s ability to detoxify herbicides, or enable transport to a site within the plant where the herbicide is not lethal.
Each time the herbicide is applied, susceptible plants die and those with resistance survive (Figure 16.1).

Figure 16.1 Rare genes for herbicide resistance may pre-exist in a weed population. The proportion of resistant to susceptible weeds will change under selection pressure.
The initial frequency of plants with pre-existing resistance is usually very low. Therefore, the majority of plants in a wild weed population will be susceptible to herbicides effective on that species (Figure 16.1).
Persistent use of herbicides with the same MOA will kill the susceptible portion of the population, resulting in the gradual increase in the proportion of resistant individuals
This process is described as applying ‘selection pressure’. By removing susceptible plants from the population, plants that can survive application of the herbicide (at the given rate) are selected.
Importation of resistance
It is possible that resistance may not be present in the population initially, but is introduced as a weed contaminant in crop seed or fodder, on machinery or on/in animals. This is particularly important for ‘rarer’ forms of resistance such as glyphosate resistance.
|
Natural dispersal
Weed seeds can also be spread by wind and water; for example, species such as sowthistle and fleabane can be spread up to 20 m by the wind. Pollen can also be dispersed great distances although the percentage remaining viable at distances greater than 10 m is low. Floodwater also has the potential to move a wide range of weed seeds over large distances. Dispersal by these mechanisms can increase the speed of development of herbicide resistance if integrated weed management (IWM) is not implemented.
Factors influencing the development of resistance
Herbicide resistance is normally present in some individual plants of weed populations before herbicides are first applied. Several factors may affect the number of seasons in which a herbicide is applied before the general population becomes resistant. These are:
- initial frequency of resistance gene(s);
- mode-of-action group of the herbicide being applied;
- herbicide efficacy;
- the size of the weed population;
- the proportion of the weed population treated; and
- weed biological factors.
Initial frequency of resistance gene
The frequency of individuals able to survive a herbicide application varies for different herbicides.
For example, high initial resistance in three untreated annual ryegrass (Lolium rigidum) populations explains the rapid evolution of resistance to Group B herbicides in this weed species once the herbicides are used. This is due to the high numbers of individual plants able to survive and reproduce after herbicide application.
For other herbicides the initial frequency may be as high as one plant in every 10,000 or as low as one plant in every billion (Table 16.1). Where initial frequencies of resistance are higher, fewer herbicide applications are necessary for resistance to develop.
Table 16.1 Initial frequency of resistant individuals of annual ryegrass, estimated by modelling (Diggle et al., unpublished data).
Group A |
Group B |
Group M |
|
Estimated initial frequency |
1 plant in |
1 plant in |
1 plant in |
|
Mode-of-action group of the herbicide being applied
The number of times a herbicide can be applied prior to a weed population developing resistance will vary. Some rules of thumb regarding the number of years of effective herbicide application before resistance evolves, according to MOA group of the herbicide being used, are given in Table 16.2.
In the case of herbicides such as triazines (Group C) and dinitroanilines (Group D), the frequency of individuals with a resistant gene enabling plants to survive the herbicide application is lower than for Group A and B herbicides. A longer period of exposure to the selection pressure (10 or more years of application) is required for weed populations to become resistant to these herbicides.
Table 16.2 Number of years of herbicide application before resistance evolves (based on Preston et al. 1999)
| Herbicide group | Years of application | Herbicide resistance risk |
| A | 6–8 | High |
| B | 4 | High |
| C | 10–15 | Medium |
| D | 10–15 | Medium |
| F | 10 | Medium |
| I | Not known | Low |
| L | >15 | Low |
| M | 15 | Low |
Herbicide efficacy
The level of kill or efficacy of the herbicide used will also affect resistance development. Highly efficacious herbicides exert strong resistance selection pressure. Modelling by Powles et al. (1997) showed that herbicides resulting in 95% weed control increased the rate of resistance development to a greater extent than herbicides resulting in 80% weed control.
The size of the weed population
The larger the number of weeds present, the greater the likelihood that there will be individuals with resistance to the herbicide within the population.
A useful analogy to understand the influence of size of weed population is the presence of white-flowered individuals in a Paterson’s curse (Echium plantagineum) population. In a small population white-flowered individuals are unlikely to be present, but their numbers increase as population density increases. The gene controlling white flower colour is rare but, importantly, is already present in the population.
Similarly, genes controlling herbicide resistance are relatively rare. As with white-flowered Paterson’s curse, the likelihood of resistant individuals being present will increase with increasing weed population. Unfortunately, unlike the white-flowered Paterson’s curse, resistant plants look exactly the same as susceptible plants and will not be detected until they survive herbicide application.
The proportion of the weed population treated
If a greater proportion of the weed population is treated with the herbicide, more susceptible individuals will be killed and the selection pressure will increase. This might occur where multiple applications of the herbicide are made in one season, for example the use of glyphosate to control barnyard grass in northern summer fallows. It could also occur where a herbicide is applied late after more weeds have emerged, for example a late post-emergent application of metsulfuron to control broadleaf weeds in winter cereals. Herbicides with a long persistence in the soil such as chlorsulfuron (used as a preemergent herbicide on light-textured alkaline soils) can also increase the selection pressure.
Weed biological factors
There are a number of key biological factors that will influence the number of years of herbicide application necessary before a weed population becomes resistant. These include:
Seedbank life. Resistance is slower to appear in weed species that have higher levels of seed dormancy. While the seed produced after each application of herbicide may contain a higher proportion of resistant individuals, susceptible seed from the seedbank will dilute resistance levels;
Fitness of resistant biotypes. In some instances herbicide resistant weeds may be less vigorous than susceptible plants of the same species. The ability of the weed to compete with other plants and set seed may therefore be reduced. Development of resistance may be slower where there is a significant fitness penalty associated with the resistance mechanism. For example, triazine (e.g. atrazine) resistance has a fitness penalty because the resistance mechanism involves a mutation in photosynthesis, the engine for plant growth. Hence, triazine-tolerant canola varieties have a lower yield potential compared with conventional lines;
Seed production. The greater the number of seeds produced by a resistant plant, the greater the number of resistant plants that will need to be controlled in the following year. Annual ryegrass can produce up to 80,000 seeds/m2 and wild radish and charlock around 30,000 seeds/m2;
Importation of resistance. It is possible for resistance to be introduced into a weed population, although the impact it has will depend on the weed numbers involved. Introduction can be the result of various seed dispersal mechanisms (e.g. resistant seed in stock feeds, hay, crop seed, machinery and soil) or animal movement. This is particularly important with rare forms such as glyphosate resistance; and
Chance. The distribution of resistant individuals within a population is not uniform. On average, all ryegrass populations start off with about 1 plant in 17,000 with resistance to Group B herbicides. In reality, some populations have 1 plant in 8,000, and others 1 in 100,000, purely as a function of chance.
Go to Activity 16-1 |
Herbicide resistance in Australia
Throughout the world herbicide resistance is an increasing problem. Information compiled by Dr Ian Heap at: www.weedscience.org/in.asp provides details of worldwide and Australian herbicide resistant weeds.
Worldwide, more weed species have developed resistance to Group B herbicides than to any other MOA group. A large number of grass (Table 8.3) and broadleaf (Table 8.4) weed species have populations which have been confirmed to be resistant to a range of herbicides across Australia.
|
Weed species at risk
A wide range of crop weeds in Australia have populations confirmed to be resistant to a range of herbicide MOA groups.
Global examples of herbicide resistance are presented in Table 8.5. Although these weeds are present in Australia, to date no populations of the herbicide resistances shown in this table have been reported here in these species. In Australia’s northern grain region, populations of 10 species of weeds have been confirmed as herbicide resistant, and more have been identified as at risk of developing resistance, particularly to glyphosate. Apart from common sowthistle and summer grasses, the weed threats differ across the northern region according to the farming system used.
It is mostly winter weeds that are at greatest risk in central New South Wales, whereas a mix of both summer and winter weeds are at risk in northern New South Wales and southern Queensland. Summer weeds are at the greatest risk of developing resistance in central Queensland (Walker et al. 2004).
Summer weeds include sweet summer grass, barnyard grass, liverseed grass, common sowthistle and parthenium weed. The risk for winter weeds is mainly expansion of current known problems such as glyphosate resistance in annual ryegrass, Group B resistance in brassica weeds and Group A resistance in wild oats. Glyphosate and Group B resistance in wild oats are also considered a risk.
The extensive use of trifluralin (Group D) in no-till farming systems in southern Australia is considered a high risk for resistance in annual ryegrass. Shepherd’s purse and brome grass are also at risk of developing Group B resistance.
While there are no populations of annual ryegrass identified as resistant to paraquat in Australia, there is resistance to paraquat in South African vineyards. Paraquat (Group L) resistance was also confirmed in weed populations in Conyza bonariensis (1989) in Egypt and Poa annua (1981) in the United Kingdom (Heap 2006).
Table 16.3 Known populations of herbicide resistant grass weeds in Australia (compiled by Stewart, 2005).
| Weed species | Herbicide group | Example herbicide | States with confirmed resistant populations | |||||
| WA | SA | Vic | NSW | Tas | Qld | |||
| Annual ryegrass (Lolium rigidum) |
A - ‘fops’ A - ‘dims’ B - sulfonylureas B - imidazolinones C - triazines C - substituted ureas D - dinitroanilines F - triazoles M - glycines | diclofop
sethoxydim shlorsulfuron imazapic, imazapyr simazine, atrazine diuron trifluralin amitrole glyphosate | X
X X X X X X X | X X X X X X X X X | X X X X X X X X | X X X X X X | X X | |
| Barley grass (Hordeum leporinum;Hordeum glaucum) | A - ‘fops’ A - ‘dims’ L – bipyridiliums B – sulfonylureas | haloxyfop, fluazifop
sethoxydim paraquat sulfosulfuron, sulfometuron |
| X X X | X
X | X |
X | |
| Broome grass (Bromus diandrus) | A – ‘fops’ | haloxyfop | X | |||||
| Barnyard grass (Echinochloa crusgalli) | C – triazines | atrazine | X | |||||
| Broome grass (Bromus rigidus) | A – ‘fops’ | quizalofop | X | |||||
| Giant Parramatta grass (Sporobolus indicus) | J – alkanoic acids | fluproponate | X | |||||
| Large crabgrass (Digitaria sanguinalis) | A – ‘fops’ B – imidazolinones | fluazifop; haloxyfop
imazethapyr | X | X
X | ||||
| Liverseed grass) (Urochloa panicoides) | C – triazines | atrazine | X | |||||
| Paradoxa grass (Phalaris paradoxa) | A – ‘fops’
A – ‘dims’ | fluazifop
sethoxydim | X
X | |||||
| Serrated tussock (Nasella trichotoma) | J – alkanoic acids | fluproponate | X | |||||
| Silver grass (Vulpia spp.) | L – bipyridiliums | paraquat | X | |||||
| Wild oat (Avena spp.) | A – ‘fops’
A – ‘dims’ B – sulfonylureas K – aminopropionates | diclofop,
tralkoxydim iodosulfuron-methyl-Na flamprop-methyl | X X | X X X | X
X | X X X | X X | |
Table 16.4 Known populations of herbicide resistant broadleaf weeds in Australia (compiled by Stewart 2005)
| Weed species | Herbicide group | Example herbicide | States with confirmed resistant populations | |||||
| WA | SA | Vic | NSW | Tas | Qld | |||
| African turnip weed (Sisymbrium thellungi) | B – sulfonylureas | chlorsulfuron | X | |||||
| Black bindweed (Fallopia convolvulus) | B – sulfonylureas | chlorsulfuron | X | |||||
| Calomba daisy (Pentzia suffruticosa) | B – sulfonylureas | metsulfuron-methyl | X | |||||
| Capeweed (Arctotheca calendula) | L – bipyridiliums | paraquat, diquat | X | |||||
| Charlock (Sinapis arvensis) | B – sulfonylureas | chlorsulfuron | X | |||||
| Common sowthistle (Sonchus oleraceus) | B - sulfony lureas | chlorsulfuron | X | |||||
| Dense-flowered fumitory (Fumaria densiflora) | D – dinitroanilines | trifluralin | X | X | ||||
| Indian hedge mustard (Sisymbrium orientale) | B – sulfonylureas
B – sulfonamides B – imidazolinones | chlorsulfuron
metosulam imazethapyr | X X | X X X | X X | X X | ||
| Paterson’s curse / (Echium plantagineum) | B – sulfonylureas B – sulfonamides | chlorsulfuron
metosulam | X
X | X X | ||||
| Prickly lettuce (Latuca serriola) | B – sulfonylureas
B – imidazolinones | triasulfuron imazethapyr | X X | |||||
| Sand rocket (Diplotaxis tenuifolia) | B – sulfonylureas | chlorsulfuron | X | |||||
| Stinging nettle (Urtica urens) | C – triazines | simazine, atrazine | X | |||||
| Turnip weed (Rapistrum rugosum) | B – sulfonylureas | chlorsulfuron | X | X | ||||
| Wild radish (Raphanus raphanistrum) | B – sulfonyl ureas
B – sulfonamides B – imidazolinones C – triazines C – triazinones F – nicotinanalides I – phenoxies | chlorsulfuron metosulam imazapic, imazapyr simazine, atrazine metribuzin diflufenican 2,4-D | X X X X X X X | X | X X | X | X | ||
| Wild turnip (Brassica tournefortii) | B – sulfonylureas
B – sulfonamides | chlorsulfuron
metosulam | X | X
X | ||||
| Note: Collated from information presented at www.weedscience.org/in.asp and other published literature. | ||||||||
Table 16.5 Resistance watch: confirmed resistances in overseas populations of common weed species in crops (compiled by Stewart, Walker and Storrie 2005)
Current status of herbicide resistance in the Northern Grain Region
(Modified from the Northern Herbicide Resistance Reporter, September 2004)
The GRDC-funded project, ‘Detection, monitoring and management of herbicide resistance – northern region’ (UQ 138), commenced in 2001 at the University of Queensland. As one of its activities, between October 2001 and June 2004, over 600 weed populations from the northern region were collected and screened for resistance, including an additional 56 populations that were sent by farmers and agronomists for resistance testing (see Map 8.1).
Field surveys
The random surveys were conducted towards the end of the summer and winter cropping periods when weed seed was mature. Most of the sampling was from in-crop paddocks and, because glyphosate resistance poses a major threat to fallow weed control in this region, fallow paddocks were included in the sampling (Figure 16.2).
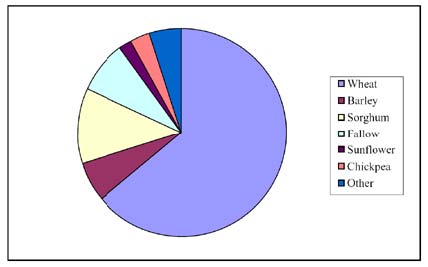
Figure 16.2 The proportion of the sites that were under a certain kind of crop or fallow regime when tested for herbicide resistant weed populations.
Seed was collected from representative samples of the most abundant mature weed species in each paddock. Wild oats accounted for 50% of the species that were collected and screened. Approximately 95% of the wild oats were Avena sterilis ssp. ludoviciana and the remaining were Avena fatua.
Another seven species accounted for 35% of the remaining total; Common sowthistle (14%), African turnip weed (4%), bladder ketmia (4%), barnyard grass (4%), turnip weed (3%), black bindweed (2%), annual ryegrass (2%), and liverseed grass (2%). The remaining 15% consisted of a large range of grass and broadleaf weeds (Figure 8.3). The large percentage of wild oats that were sampled was primarily due to their abundance within wheat and barley paddocks but could also have been slightly biased by the fact that wild oats exist as different aged cohorts within a crop and, therefore, mature over a relatively long period. For other species the timing of a collection survey must coincide within a much narrower window in order to collect mature seed.
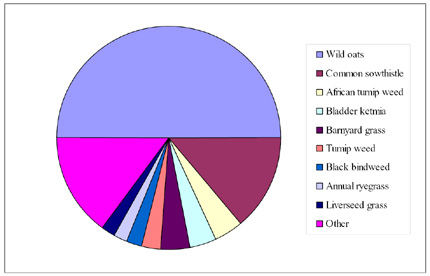
Figure 16.3 The proportion of the various weed species collected and screened for herbicide resistance status.
Screening techniques
The randomly collected wild oat populations were screened with a Petri dish bioassay called the Rothamsted Rapid Resistance Test (RRRT) that was developed by Dr Stephen Moss at the Rothamsted Research Station in the UK. This test imbibes the wild oat seed in Petri dishes, which have the Group A ‘fop’ herbicide (fenoxaprop-p-ethyl e.g. Wildcat) added to the filter papers at a very low concentration (7.5 ppm). If the wild oat seed can germinate and produce at least 1 cm of green shoot material on the herbicide solution, then this indicates resistance. A comparison is made against two reference populations, one being a known resistant population, to determine the level, if any, of herbicide resistance. A susceptible population is one that is adequately controlled or killed by the herbicide.
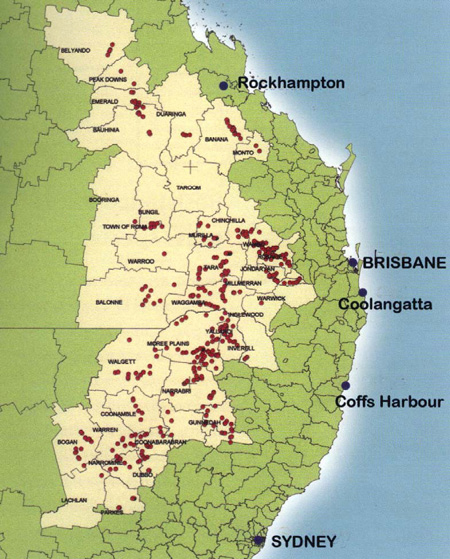
Map 16.1 The 608 weed populations tested for their herbicide resistance status came from a large number of sampling locations throughout the northern grain region of Queensland and New South Wales.
When the plant response to herbicides is absolute, interpretation of the result is relatively straightforward. That is, if 100% of plants are killed then the population is susceptible, and if 100% survive then the population is resistant. This test also classifies populations as ‘partially resistant’ and ‘strongly resistant’, but these terms have been combined to ‘resistant’ in this report and accompanying maps. The RRRT takes 14 days to complete but requires the wild oat seed to have little or no dormancy in order to obtain acceptable germination percentages. This means that the test cannot usually be conducted on freshly collected seed as this seed normally has high levels of dormancy. This necessitates the pre-requisite step of placing the wild oat seed in an incubator at a constant temperature (35°C) for 4–6 weeks until the dormancy level has declined sufficiently.
We found that the RRRT had a slight tendency to overestimate the level of resistance. That is, it would sometimes return a ‘false positive’ result, and indicate that a population had a low level of resistance or insensitivity when it was actually susceptible. For this reason, populations that were assessed as resistant were often rescreened with a standard pot test (see below) to confirm results.
All wild oat seed sent in by growers and agronomists was screened for Group A (herbicides that bring about their effect by inhibiting ACC-ase activity in sensitive plants) resistance using standard pot tests. These populations were screened with the same chemical that the farmers had used in the paddocks and failed to get satisfactory weed control with. Some populations were subjected to dose-response, pot test trials using seven application rates of ¼, ½, 1, 2, 4, 8, and 16 times the recommended rate of the herbicide. These trials were conducted in the wind tunnel laboratory with precision application equipment. All other species were tested using standard pot tests.
Resistance status of grass weeds
There were 28 wild oat populations that were resistant to Group A herbicides (Map 8.2), which is approximately 10% of all samples taken. Eleven of the 28 resistant populations were very strongly resistant and had 100% survival at 16 times the recommended application rate of the Group A herbicide.
Two of the Group A resistant wild oat populations were resistant to both ‘fop’ and ‘dim’ chemical classes, and two populations were cross resistant to the recommended rate of flam-prop-m-methyl (e.g. Mataven), a Group K herbicide.
Two populations of annual ryegrass were resistant to Group A herbicides with one population being both ‘fop’ and ‘dim’ resistant.
The glyphosate resistant (Group M) annual ryegrass populations in the Quirindi Shire were detected and confirmed by staff from the NSW DPI.
As well, one strongly resistant barnyard grass population to atrazine, a Group C herbicide, was found in the Moree Plains Shire in northern NSW.
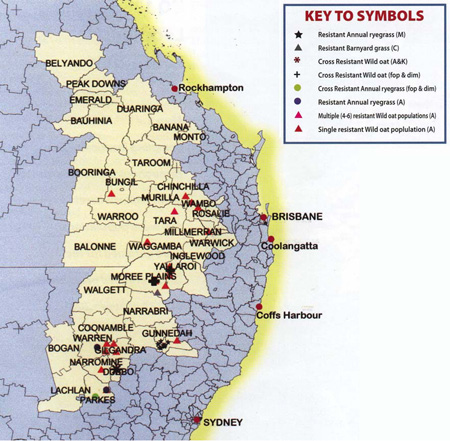
Map 16.2 The herbicide resistance status for the grass species collected in the northern grain region.
Letters or abbreviations in brackets indicate the chemical group or class of chemical that the population is resistant to.
Of the 300 wild oat populations that were tested, approximately 95% of these were Avena sterilis ssp. ludoviciana while the remaining 5% were Avena fatua.
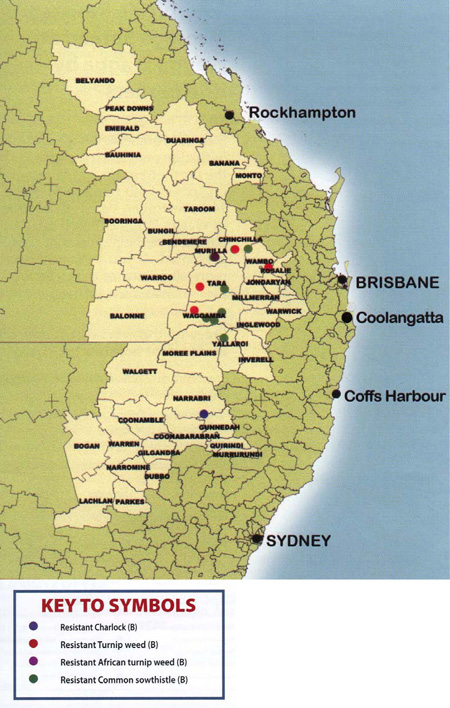
Map 16.3 Resistance status – broadleaf weeds (resistance status of broadleaf weed species that were screened as part of project UQ 138.
Letters in brackets indicate the chemical group that the population is resistant to).
Resistance status of broadleaf weeds
Four broadleaf weed species, common sowthistle, turnip weed, African turnip weed, and charlock, were found to be resistant to Group B herbicides. Most of these resistant weeds were from Queensland, predominantly in the Waggamba, Wambo and Tara shires. There were only two resistant broadleaf weed populations from New South Wales, a resistant charlock population from Narrabri shire and one resistant common sowthistle population from the Yallaroi shire. This pattern is somewhat different to what had been detected in earlier University of Queensland projects, which detected a total of 39 resistant broadleaf weed populations and 22 of these were from the Moree Plains and Yallaroi shires of New South Wales.
Mechanism of Group A resistance
The mechanism for the strongly resistant wild oat populations has been characterised through molecular biology studies, and found to be ‘target site’ resistance. Target site resistance results from a genetic change so that the site where the herbicide molecule binds or acts within the plant is changed and the herbicide is no longer recognised.
The molecular characterisation has detected mutation sites, not previously found in other wild oat populations. UQ has subsequently developed a PCR-based molecular diagnostic test for these mutations.
Summary
Prior to 1993, when the first resistance screening project was undertaken at UQ, there were no confirmed instances of herbicide resistance in the northern region. In a period of just over 10 years, a total of 110 weed populations have been confirmed as resistant (see Map 8.4). This rapid increase in the incidence of herbicide resistant weed populations is consistent with the world-wide trend (Figure 8.4) and there are presently no signs that it is about to level out.
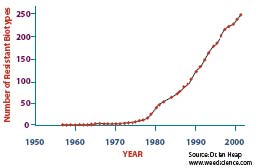
Figure 16.4 Worldwide increase in the number of herbicide resistant weed biotypes since the late 1950s
The future
History has shown that the trend for increasing herbicide resistance in Australian cropping systems is likely to continue, at least in the near future. Due to the great success of herbicides improving weed control and farmer returns over the last 30 years, non-herbicide management has been forgotten by the majority of advisors and growers.
Herbicide resistance is the impetus for the relearning of integrated weed management. Growers in more favourable climatic areas have more options available and better cash flows to fund necessary changes in management. Growers in drier areas, however, face greater challenges in managing highly variable seasonal conditions and cash flows, which determine their ability to adopt and implement change. Convincing growers to introduce changes in weed management sooner rather than later is a challenging and long-term task for all farm advisors.
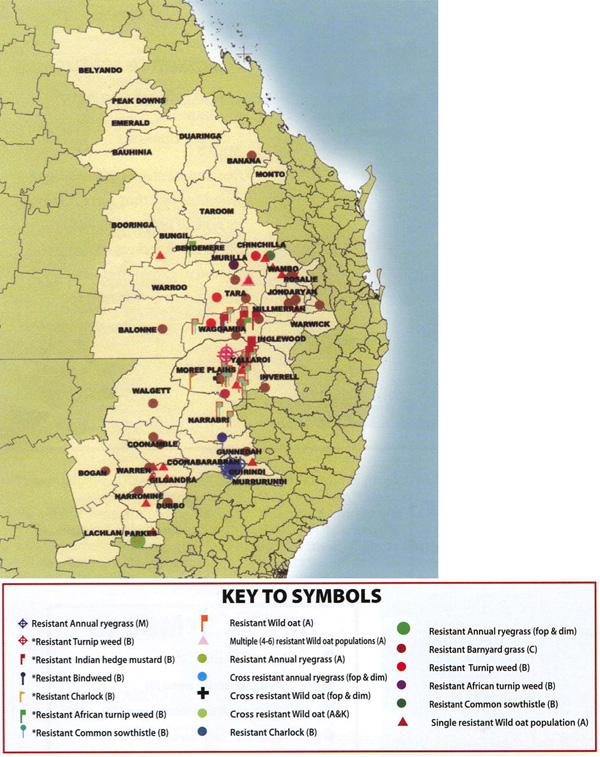
Map 16.4 Herbicide resistant weed populations in the Northern Grain Region, based on results from current and former UQ projects.
Symbols prefaced by an asterisk (*) indicate resistant populations that were detected as part of the earlier projects prior to 2001.
References
Alizadeh, HM, Preston, C and Powles, SB 1998, ‘Paraquat-resistant biotypes of Hordeum glaucum from zero-tillage wheat’, Weed Research, vol. 38, pp. 139-142.
Brown, TM (ed.) 1996, Molecular genetics and evolution of pesticide resistance, American Chemical Society, Washington, DC.
Caseley, JC, Cussans, GW & Atkin, RK, (eds.) 1991, Herbicide resistance in weeds and crops, Butterworth-Heinemann, Oxford.
Denholm, I, Devonshire, AL & Hollomon, DW 1992, Resistance ‘91: Achievements and developments in combating pesticide resistance, Elsevier Applied Science, London.
Diggle, AJ and Neve, P 2001, ‘The population dynamics and genetics of herbicide resistance – a modeling approach’, in Herbicide resistance and world grains, CRC Press Inc., Boca Raton, USA: pp. 6-99.
Gill, GS 1995, ‘Development of herbicide resistance in annual ryegrass populations (Lolium rigidum Gaud.) in the cropping belt of Western Australia’, Australian Journal of Experimental Agriculture, vol. 35, pp. 67-72.
Green, MB, LeBaron, HM and Moberg, WK (eds) 1990, Managing resistance to agrochemicals: From fundamental research to practical strategies, American Chemical Society, Washington, D.C.
Heap, I 2006, Criteria for Confirmation of Herbicide-Resistant Weeds, www.weedscience.com
Llewellyn, RS and Powles, SB 2001, ‘High levels of herbicide resistance in rigid ryegrass (Lolium rigidum) in the wheat belt of Western Australia’, Weed Technology, vol. 15, pp. 242-248.
Powles, SB & Holtum, JAM, (eds) 1994, Herbicide resistance in plants: Biology and biochemistry, Lewis Publishers, Boca Raton, Fl.
Powles, SB & Shaner, DL, (eds) 2001, Herbicide resistance and world grains, CRC Press, Boca Raton, Fl.
Powles, SB, Preston, C, Bryan, IB and Jutsum, AR 1997, ‘Herbicide resistance: Impact and management’, Advances in Agronomy, vol. 58, pp. 57-84.
Preston, C 2004, ‘Herbicide resistance and its management’, in Proceedings of 2004 South Australian GRDC Crop Updates, (www.grdc.com.au/growers/res_upd/south/s04s/preston2.htm).
Preston, C & Mallory-Smith, CA 2001, ‘Biochemical mechanisms, inheritance, and molecular genetics of herbicide resistance in weeds’ in SB Powles & DL Shaner (eds), Herbicide resistance and world grains, CRC Press, Boca Raton, Fl, pp. 23-60.
Preston, C, Roush, RT and Powles, SB 1999, ‘Herbicide resistance in weeds of southern Australia: why are we the worst in the world?’, in AC Bishop, M. Boersma and CD Barnes (eds) 12th Australian Weeds Conference: Papers and Proceedings. Tasmanian Weed Society, Devonport, Australia, pp. 454-459.
Purba, E, Preston, C & Powles, SB 1993, ‘Inheritance of bipyridyl herbicide resistance in Arctotheca calendula and Hordeum leporinum’, Theoretical and Applied Genetics, vol. 87, pp. 598-602.
Storrie, AM and Cook, AS 2002, ‘Glyphosate resistance in northern NSW – a growing concern, in Proceedings 13th Australian Weeds Conference, Perth, Western Australia, pp. 601–604.
Tucker, ES & Powles, SB 1988, ‘Occurrence and distribution in south-eastern Australia of barleygrass (Hordeum glaucum Steud.) resistant to paraquat’, Plant Protection Quarterly, vol. 31, pp. 19-21.
Walker, S, Widderick, M, Storrie, A and Osten, V 2004, ‘Preventing glyphosate resistance in weeds of the northern grain region’, in Weed management: balancing people, planet, profit, Weed Society of New South Wales, Sydney, Australia. pp. 428–431.